How an Oil Refinery Works

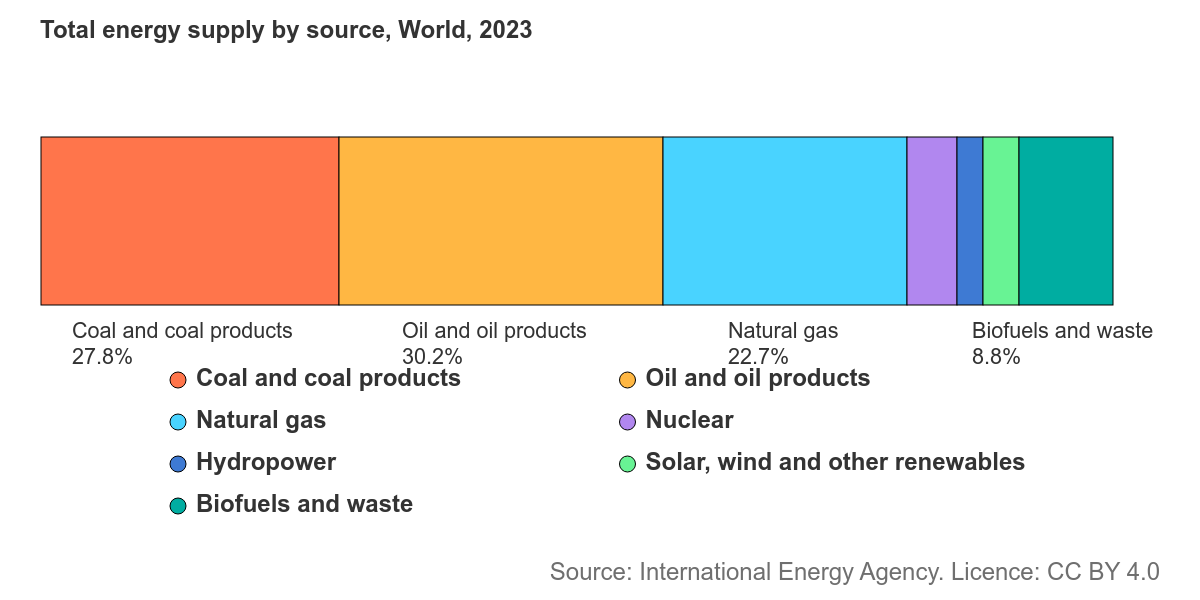
Though wind and solar continue to carve out larger and larger shares of world energy supply, the modern world still runs on petroleum, and will continue to do so for the foreseeable future. The world consumes over 100 million barrels of oil a day. As of 2023, oil was responsible for 30% of all energy use worldwide, higher than any other energy source (though its share has been gradually falling). In chemical manufacturing, petroleum is even more critical: an astounding 90% of chemical feedstocks are derived from oil or gas. Virtually all plastic comes from chemicals extracted from oil or gas, and petrochemicals are used to produce everything from lubricants to paint to plywood to synthetic fabrics to fertilizer.
Our enormous consumption of petroleum is made possible by oil refineries. When oil comes out of the ground, it’s a complex mixture of thousands of different chemicals. Oil refineries take in this mixture and process it, turning it into chemicals we can actually use. Because of the scale of worldwide petroleum consumption, oil refineries are some of the largest industrial facilities in the world. A large oil refinery will occupy thousands of acres and cost billions of dollars to construct, ultimately refining hundreds of thousands of barrels of oil each day.
Crude oil basics
Oil is a liquid produced from decomposing organic materials, mostly plankton and algae that died and sank to the bottom of ancient oceans. This dead organic matter gradually got covered with sediment, and over millions of years it transformed into crude oil. Crude oil is a mixture of thousands of different chemicals, most of which are hydrocarbons: molecules that are various arrangements of carbon and hydrogen atoms. The molecules in crude oil range from the simple, such as propane (three carbons and eight hydrogens) and butane (four carbons and ten hydrogens) to the complex — some asphaltene molecules in crude oil can contain thousands of individual atoms.1
Crude oils extracted from different parts of the Earth will have different mixtures of hydrocarbons and other molecules, which has given rise to a sort of crude oil taxonomy. “Heavy” crude oils, found in places like Canada’s oil sands, will have more heavy molecules, while “light” crude oils found in places like Saudi Arabia’s Ghawar field will have more light molecules. “Sweet” crudes, like the crudes extracted from the Brent oil field in the North Sea, have lower sulfur content, while “sour crudes,” like some of the crudes extracted from the Gulf of Mexico, have greater sulfur content.
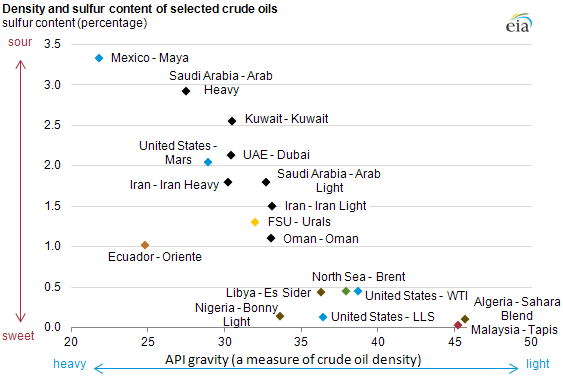
The job of an oil refinery is to process this mixture of hydrocarbons and other molecules: separating the mixture into individual chemicals or groups of chemicals, and using various chemical reactions to change low-value chemicals into more valuable, useful ones.
Refinery basics
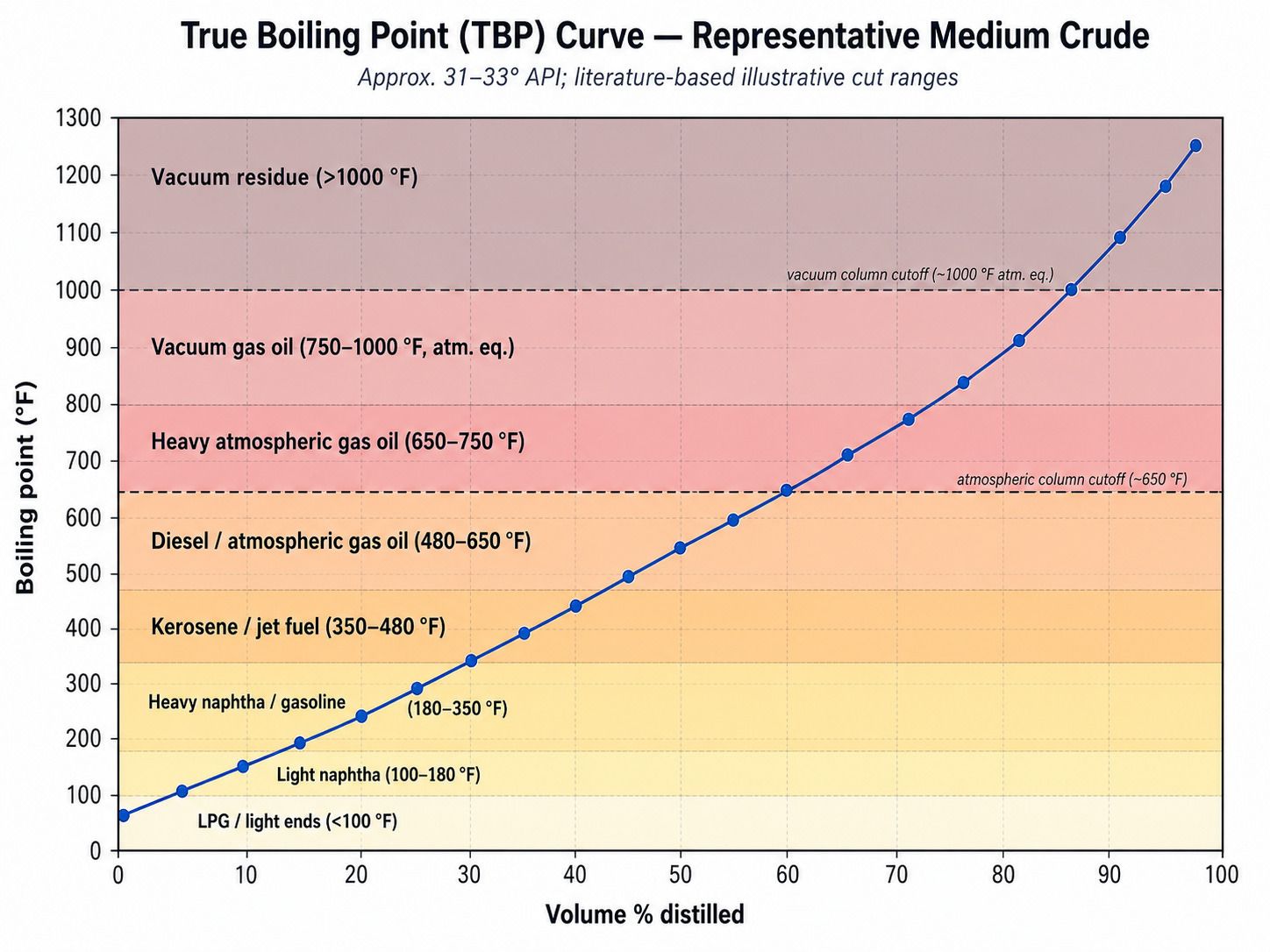
A refinery makes use of several different methods to separate and process crude oil, but the most important process of all is probably distilling. Different molecules within crude oil boil at different temperatures, and condense back into liquid at different temperatures. Smaller, lighter molecules boil and condense at lower temperatures, while larger and heavier molecules boil and condense at higher temperatures. You can describe this range of boiling points with a distillation curve, which shows what fraction of the crude oil boils at different temperatures. In the example curve below, we can see that at about 350°C half the crude has boiled off, and at 525°C about 80% of the crude has boiled off. Different crude oils will have slightly different distillation curves, depending on the proportion of different molecules within them.
Substances derived from crude oil are often mixtures of chemicals defined by their range of boiling points. Gasoline, for instance, isn’t just one chemical: it’s a mixture of hydrocarbons, mostly molecules with between four and 12 carbon atoms. The EIA defines finished gasoline as “having a boiling range of 122 to 158 degrees Fahrenheit at the 10 percent recovery point to 365 to 374 degrees Fahrenheit at the 90 percent recovery point.”2
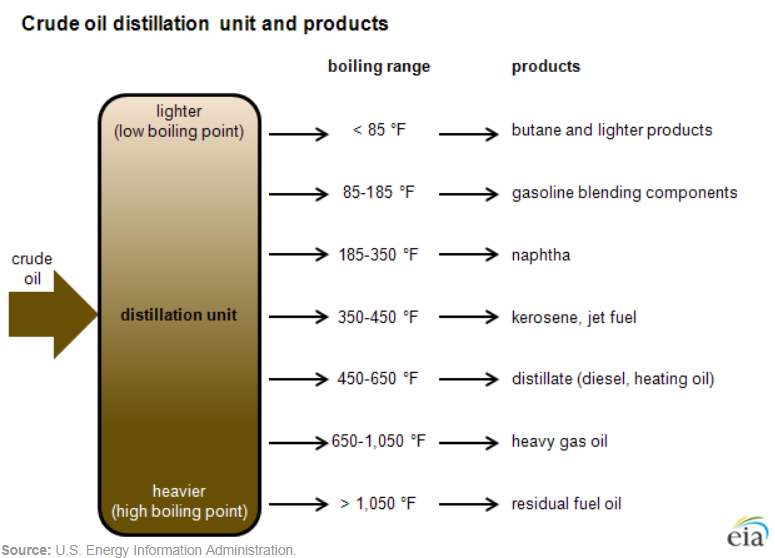
Oil refineries can use this range of boiling and condensation to separate crude oil into different groups of chemicals, or fractions, using a distillation column. When crude oil enters a refinery, the salt gets removed from it, and it’s then heated to around 650-750°F, which turns most of the oil into a vapor. The vapor is then fed into a tall column containing trays at different heights, each filled with liquid. As the hot vapor rises through the column, at each tray it passes through the liquid, which cools it slightly. When the vapor cools enough, it condenses back into liquid. The heaviest molecules with the highest boiling points condense first, at the bottom of the column, while the lighter ones condense last, at the top. The very lightest molecules don’t condense at all: they exit the top of the column while remaining a gas. At the same time, the very heaviest molecules remain a liquid the entire time, and exit the bottom of the column. Thus, different molecules of different weights can be separated out.
Essentially every oil refinery first distills crude oil into various fractions in a distillation column, though the exact fractions separated might vary from refinery to refinery. Because this distillation is done at atmospheric pressure, this first step in the refining process is referred to as “atmospheric distillation.” The simplest refineries might only do atmospheric distillation, but most refineries will then send these various fractions along for further processing. There are a LOT of processes that a refinery might use, depending on what it’s designed to produce, so we’ll just look at some of the most widely used ones.
The gas that comes out of the top of atmospheric distillation will be a mixture of several different light molecules — propane, methane, butane, isobutane (butane with a slightly different molecular arrangement) and so on. To separate this mixture into its component gases, a refinery can send it to a gas plant, which contains a series of distillation columns designed to condense various substances out of the mixture. So gas might flow through a “debutanizing tower” to separate butane, propane and lighter gasses from the rest of the mixture; the butane-and-lighter gasses might then be sent to a “depropanizing tower” to separate the propane from the butane.3
While light gases come out of the top of a distillation column, heavy liquids come out the bottom. The very heaviest molecules, which emerge from distillation without ever having evaporated at all, are known as residuals. Many of the heavier molecules aren’t particularly valuable by themselves, and thus one of the most important functions of a refinery is cracking — splitting heavy fractions, such as heavy fuel oil, into lighter, more valuable ones such as gasoline.
Cracking was invented in the early 20th century as a way to extract more gasoline from a barrel of crude oil to meet rising demand from car usage. Over the years cracking methods have evolved, and today most refineries use some flavor of catalytic cracking (or “cat cracking”). In catalytic cracking, the heavy fractions from atmospheric distillation are mixed with a catalyst (a material designed to speed up chemical reactions) and subjected to heat and pressure, splitting the heavy molecules into lighter ones. The catalyst is then separated from the mixture using a cyclonic separator — essentially, the mixture is spun around, separating out the heavier catalyst from the rest of the mixture — cleaned, and reused, while the now-cracked (and therefore vapor-izable) oil is sent to another distillation column which splits it into various fractions.
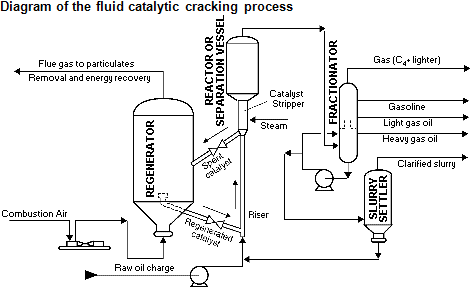
Most catalytic cracking is fluid catalytic cracking, which uses a sand-like catalyst that behaves as a fluid when mixed with the heavy fractions. Different companies have developed different fluid catalytic cracking processes, and different refineries might use multiple catalytic crackers in different parts of the process.
Catalytic crackers are designed to encourage the chemical reactions that break apart heavy hydrocarbons, but these reactions can also occur within the distillation column if the heat is high enough. Because cracking is disruptive to the distillation process, refineries limit the temperature in atmospheric distillation to around 650-750°F. This leaves behind a mixture of heavy, unboiled hydrocarbons at the bottom of the column. It would be useful to further separate this mixture into different fractions so that it could be reclaimed, but atmospheric distillation can’t do that without raising the temperature to the point where cracking starts to occur.
The solution is to send this mixture to another distillation column that’s kept at very low pressure, near vacuum — this process is thus known as vacuum distillation or vacuum flashing. Lower pressure means lower boiling points, allowing the heavy fractions to be distilled without heating them to the point where cracking starts to occur.
Some of the heavy fractions that come out of vacuum distillation might be sent directly to a catalytic cracking unit to split them into lighter ones. But the very heaviest molecules that come out of the bottom of the vacuum distillation column aren’t suitable for catalytic cracking — many of them contain heavy metals that would poison the catalyst, and the chemical reactions of these molecules tend to produce coke (a carbon-rich solid), which would gum up the catalyst. Because it’s useful to crack these very heavy molecules, some refineries will use thermal cracking processes, which use heat to split molecules apart. Cokers are thermal crackers that use heat to crack the heaviest molecules into lighter ones and coke. The lighter molecules are sent to a distillation column to be separated; the coke can be burned as fuel, or as a manufacturing input (the electrodes used in aluminum smelting, for instance, are made from coke). Another type of thermal cracking, visbreaking (short for viscosity breaking), is used to crack some molecules and reduce the viscosity of the remaining fractions.
Besides cracking, a refinery might employ a variety of other processes to modify the chemical structure of various molecules. Catalytic reforming takes the naphtha fraction (the part of the crude oil with a boiling point between ~122°F and ~400°F) and exposes it to heat and pressure in the presence of a catalyst to produce a new mixture of chemicals called reformate that is used to make gasoline. Isomerization processes take various molecules, such as butane, and modify their physical arrangement to produce isomers – molecules with identical chemical formulas but different structural arrangements. Hydrotreating reacts various crude oil fractions with hydrogen in the presence of a catalyst to remove impurities and improve their quality. (Hydrotreating can be done on its own, but it’s also often combined with other processes. Hydrocracking combines hydrotreating with catalytic cracking, and residue hydroconversion combines hydrotreating with thermal cracking.)
To store the various inputs and outputs of these processes, oil refineries also have huge numbers of storage tanks called tank farms, which are capable of storing millions of gallons of various liquids. Gases like propane and butane will typically be stored as pressurized liquids, either in above-ground tanks or in underground caverns or salt domes.
Chevron’s Richmond refinery
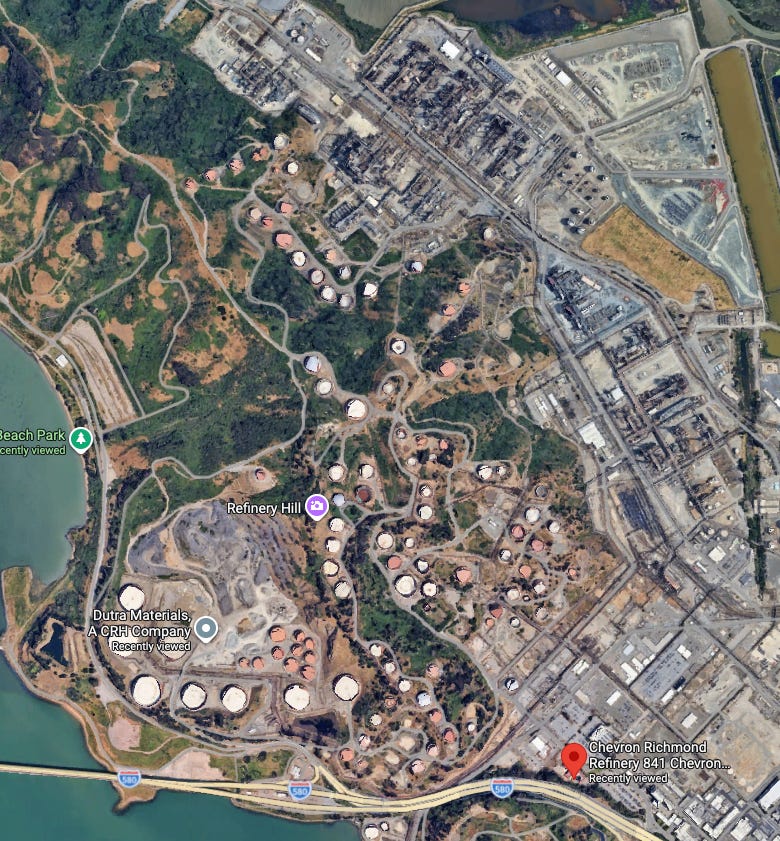
To get a sense of how these various processes might be arranged, we can look at how they’re implemented in an actual refinery. The map below shows Chevron’s Richmond, California refinery, a moderately large refinery capable of processing about a quarter million barrels of crude oil a day. The tank farm occupies the south half of the site, while the processing area wraps around the north and east.
The chart below shows the daily capacity of various processes at the refinery.
We can see that Chevron Richmond has many of the processes that we described above: in addition to ~257,000 barrels of atmospheric distillation, it has ~123,000 barrels of vacuum distillation, ~90,000 barrels of catalytic cracking, and ~71,000 barrels of catalytic reforming. (Chevron Richmond doesn’t have any coking capacity, but Chevron’s slightly larger El Segundo refinery in Los Angeles does.)
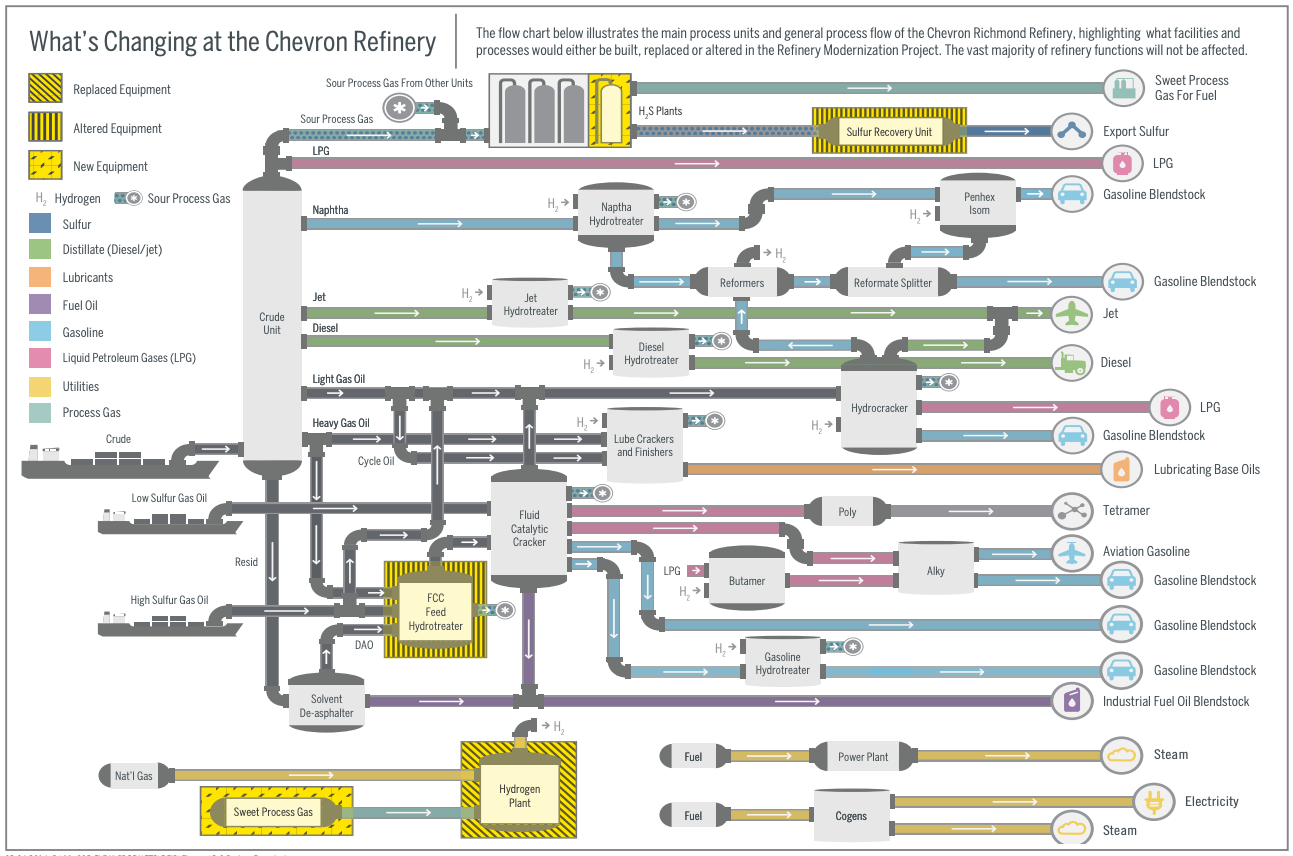
To see how these processes are actually arranged, we can look at a process flow diagram for the refinery. (This diagram is available because several years ago Chevron extensively modified this refinery, which required them to submit a very detailed environmental impact report to comply with California’s environmental quality laws.)
We can see that the refining process starts with atmospheric distillation (though the refinery also processes some heavy gas oil that can skip the distillation process), which separates the crude into various fractions. These fractions then get routed to various other processes. The light gas gets sent to the gas plant, while the naphtha gets sent to hydrotreating, catalytic reforming, and isomerization. Jet fuel and diesel fuel are sent to their own hydrotreating processes, and the heavier fractions get sent to various catalytic cracking processes. The output of all these processes is various crude oil products: heavy fuel oil, diesel, jet fuel, lubricants, and, of course, gasoline.
A broader look at oil refining capacity
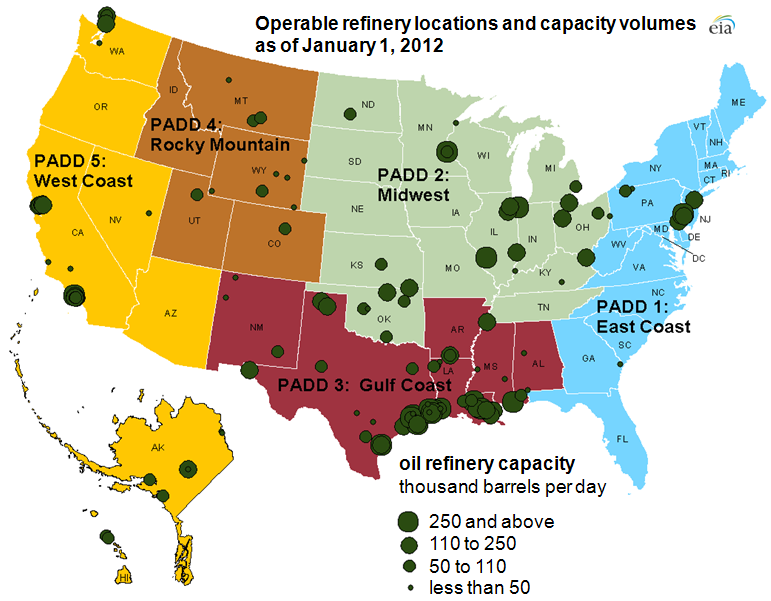
Chevron Richmond is just one of 132 operable oil refineries in the U.S., which collectively can refine over 18 million barrels of crude oil each day. The location of these refineries is highly concentrated: most of them are on the Gulf Coast of Texas and Louisiana, with other clusters in New Jersey, the Midwest, and in California.
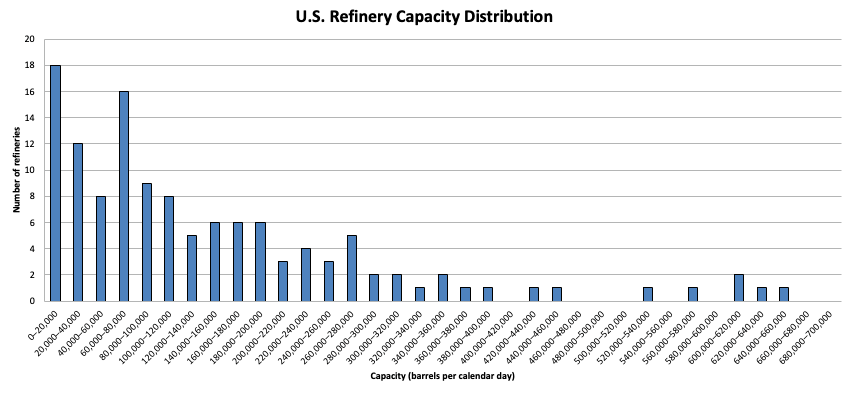
If we look at the distribution of refinery capacity we can see that Chevron Richmond is on the larger side, but far from the largest. Around a fifth of US refineries are roughly as large or larger than Chevron Richmond. Six US refineries are more than twice as large, with the capacity to refine more than half a million barrels a day. And some refineries around the world are even bigger: the Jamnagar refinery in India, the world’s largest refinery by raw capacity, can refine 1.4 million barrels of crude per day.
But looking at capacity in barrels per day (which is essentially atmospheric distillation capacity) only tells part of the story. As we noted, different refineries will have different processing equipment installed depending on what they’re designed to produce. Simple refineries will have little more than atmospheric distillation, while more complex ones will employ long sequences of processes to produce a wide range of highly refined products. The chart below shows the collective US refining capacity of various processes.
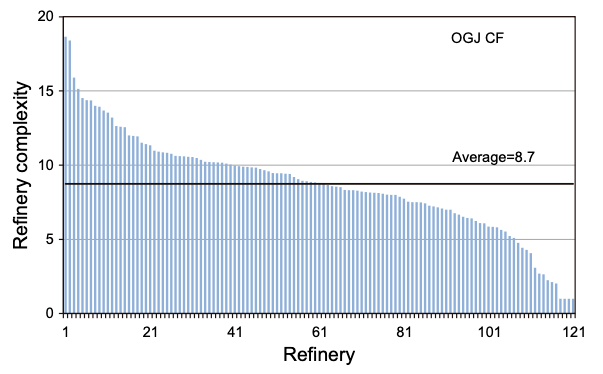
We can look at the relative complexity of different US refineries using the Nelson Complexity Index, which is intended to measure how complex a refinery is. The index is constructed by taking each process a refinery employs, and multiplying its refining capacity by a “complexity factor” that compares the cost of that process to atmospheric distillation, and then dividing by the refinery’s atmospheric distillation capacity. So a refinery that has 100,000 barrels of atmospheric distillation capacity (complexity factor of 1) and 50,000 barrels of vacuum distillation capacity (complexity factor of 2) would have a Complexity Index of 1 + 2 * 50,000 / 100,000 = 2. If it then added 25,000 barrels of catalytic cracking capacity (complexity factor of 6), its Complexity Index would rise to 1 + 1 + 6 * 25,000 / 100,000 = 3.5.
Most refineries in the US are fairly complex. As of 2014, less than 3% of refineries had a complexity index of 2 or less, and the average complexity index was 8.7. As of 2014 the Chevron Richmond refinery had a complexity index of 14, above average for US refineries. The Jamnagar refinery, in addition to being the world’s largest, is also particularly complex: its complexity index of 21 would make it more complex than virtually any US refinery.
Conclusion
What strikes me most about oil refining isn’t the complexity of the process — indeed, while the arrangements of various processes are often exceedingly complex, many of the processes themselves are often surprisingly simple (conceptually, at least). What strikes me is the sheer scale of it. Refining is an expensive undertaking not necessarily because the processes are so complex, but because the volume of material that has to be processed is so high. Chevron’s Richmond refinery is the size of a small city, and can process the entire contents of a Very Large Crude Carrier in a little over a week. And Richmond isn’t even a particularly large refinery: the US has 25 refineries that size or larger, and six refineries that are more than twice as large. Worldwide, it takes 400 Richmond-size refineries to keep the world fed with petroleum.
If you live in Texas or Louisiana these aspects are probably obvious to you, but most of us are able to go about our lives without ever thinking about the huge industrial machine that keeps the blood of civilization flowing. But the US consumes over 20 million barrels of oil a day, every day, and it takes a vast complex of oil refineries to make that possible.
Asphaltenes aren’t technically hydrocarbons: they consist mostly of carbon and hydrogen, but they can also incorporate other atoms, such as sulfur or heavy metals.
The recovery point is the temperature at which that fraction of the liquid has been vaporized and then collected.
Most of the gases sent to the gas plant will have no double bonds in them. Hydrocarbons without double bonds are known as saturated, because they have the maximum number of hydrogen atoms that they can, and so this type of plant is called a “sats gas plant”.